Transport Proteins in Honey Bees (Apis Mellifera)
 The bee project aims to elucidate the molecular mechanism of pesticide mixtures on the multi-drug resistance protein, P-glycoprotein, in honeybees. Bees are extremely important to agriculture and food production; however, bees populations are declining. We want to understand how pesticide mixtures could be contributing to this decline. Pesticides are already tested for bee safety before being released to the market but only one at a time. There are currently no regulations to test new chemicals in combination with existing pesticides even though mixtures are more environmentally relevant. Crop farmers often apply more than one chemical to treat for several different problems at once, for example, a fungicide together with an insecticide. In addition, beekeepers apply in-hive medications like antibiotics or miticides to treat for pests. We seek to characterize how these mixtures of pesticides and hive medication could interact with P-glycoprotein to cause toxic bioaccumulation or neurotoxicity in the bees.
The bee project aims to elucidate the molecular mechanism of pesticide mixtures on the multi-drug resistance protein, P-glycoprotein, in honeybees. Bees are extremely important to agriculture and food production; however, bees populations are declining. We want to understand how pesticide mixtures could be contributing to this decline. Pesticides are already tested for bee safety before being released to the market but only one at a time. There are currently no regulations to test new chemicals in combination with existing pesticides even though mixtures are more environmentally relevant. Crop farmers often apply more than one chemical to treat for several different problems at once, for example, a fungicide together with an insecticide. In addition, beekeepers apply in-hive medications like antibiotics or miticides to treat for pests. We seek to characterize how these mixtures of pesticides and hive medication could interact with P-glycoprotein to cause toxic bioaccumulation or neurotoxicity in the bees.
Intestinal Transport in Aquatic Organisms
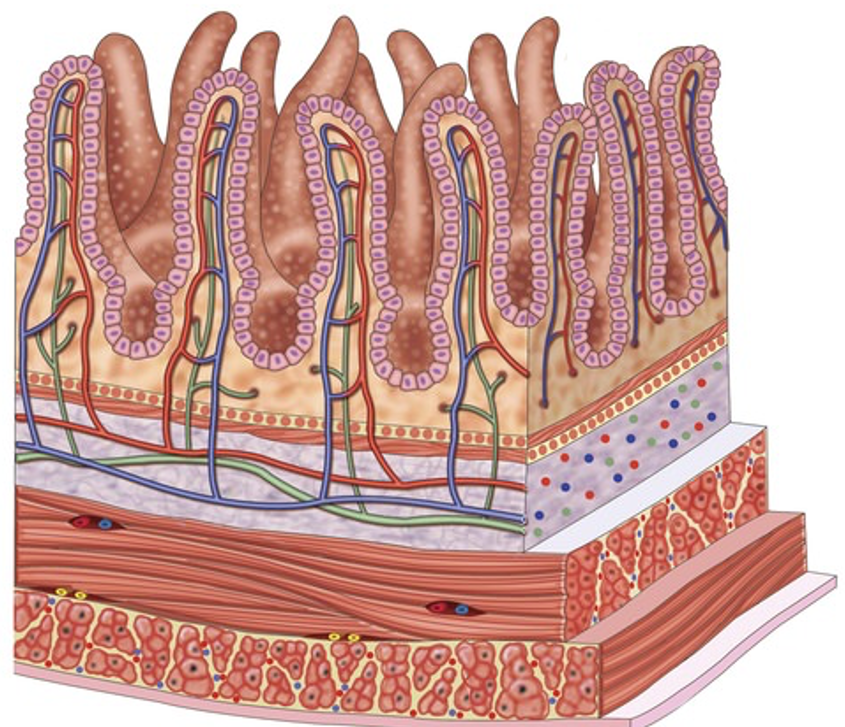 Dietary exposure is a critical exposure pathway to toxic environmental chemicals in aquatic organisms. This project will provide a perspective on current understanding of how transport proteins in intestinal epithelial tissues facilitate the pumping of chemicals through the digestive system and to other tissues in the body of aquatic organisms. The project will examine known expression patterns of various transporter protein families such as ATP Binding Cassette (ABC) proteins and Solute Carrier (SLC) proteins in select organisms. It will also discuss known compounds that can alter protein expression, act as substrates, or even inhibit the function of these proteins. Ultimately, this project will inform future research by highlighting both what is known and what isn’t known about how transport proteins can process environmental toxicants ingested by aquatic organisms.
Dietary exposure is a critical exposure pathway to toxic environmental chemicals in aquatic organisms. This project will provide a perspective on current understanding of how transport proteins in intestinal epithelial tissues facilitate the pumping of chemicals through the digestive system and to other tissues in the body of aquatic organisms. The project will examine known expression patterns of various transporter protein families such as ATP Binding Cassette (ABC) proteins and Solute Carrier (SLC) proteins in select organisms. It will also discuss known compounds that can alter protein expression, act as substrates, or even inhibit the function of these proteins. Ultimately, this project will inform future research by highlighting both what is known and what isn’t known about how transport proteins can process environmental toxicants ingested by aquatic organisms.
Vacuolar Sequestration of Compounds in Plant Cells

Plant cells are generally more static and rigid than other kinds of eukaryotic cell and must tightly regulate the movement and concentration of substances within them in a manner different than in animals or fungi. Plants use an inordinately large number of transport proteins for this job, located in the plasma membrane of the cell and in the membranes of cell compartments. The vacuole is often the largest compartment of the cell and is used to store water, regulate turgidity and sequester substances for the cell. The membrane separating the cytoplasm from the vacuolar compartment is called the tonoplast.
One group of compounds that moves from the cytoplasm into the vacuole is glycoslyated monolignols, which are precursors of the lignin polymer responsible for much of the strength in plant cell walls. It is currently unknown which kind of transport protein is responsible for transporting these compounds into the vacuole or whether this mechanism is consistent across tissues and species. The purpose for this sequestering is also currently unknown as well as how they exit the vacuole for transport to the cell wall. We are investigating these questions using purified isolated plant vacuoles and assaying their uptake under conditions of exposure to various pharmacological inhibitors. Future projects with plant transporters may involve the sequestration of pesticides and other environmental chemicals into the vacuole.
Chemical-Transporter Interaction Database
There are currently very few resources which provide a comprehensive view of how chemicals interact with transporter proteins. No comprehensive data sources currently exist to provide detailed information about interactions across different organisms or how chemicals encountered in the environment may affect transporter function. The Nicklisch Lab is actively involved in compiling data scattered from across the literature into an accessible and understandable data source for researchers, medical personnel and the public alike. We are particularly interested in classifying chemical-transporter interactions to find broad trends using kinetic, gene expression and drug-drug interaction data.
